Dr. Gregori M. Kurtzman discusses the benefits of laser implementation

Endodontic success is predicated on the ability to debride and clean the canal system. That canal system is a complex array of accessory and lateral canals, fins, and other anatomical areas inaccessible to endodontic files (Figure 1). As practitioners, we are able to clean the main canals with files, either hand or rotary, but cannot mechanically remove pulpal tissue and debris from the canal anatomy adjacent to the main canals. Treatment success requires elimination of the pulpal tissue and associated bacteria from this anatomy so that it can be sealed during the obturation phase of treatment. As only one thing can occupy a space at a time, obturation material cannot fill areas still occupied by pulpal tissue. Success is dependent on disinfection and debridement of the canal system so that it may be sealed during obturation. Irrigation has long been accepted as a key factor of treatment to achieve those goals.
Yet complete clearing of residual bacteria, especially in the apical portion of the canal system, has been difficult to achieve with traditional methods using even sodium hypochlorite (NaOCL) solutions (Figure 2). Studies have demonstrated that addition of an Er:YAG laser to activate the irrigation solution greatly enhances not only greatly enhances the efficiency of the irrigation solutions advocated (NaOCL and EDTA,) but also improves disinfection of the canal system, clearing accessory so that it may be sealed during obturation (Figures 3-4).

Irrigation: the key to endodontic success
Although instrumentation with files is important to enlarging the canals and readies them to be obturated, debris consisting of pulpal tissue and associated bacteria is not effectively removed by files. Irrigation with an appropriate solution is required to remove that debris from the canal walls. NaOCL is still the accepted irrigant due to its tissue-dissolving ability and antibacterial nature. Yet it cannot effectively reach far beyond the main canals to remove the residual tissue. Tissue dissolution can be enhanced to more effectively remove pulpal tissue/bacteria and also reach further into the accessory anatomy to allow better sealing of the canal system improving treatment success.
Smear layer within the canal system plays a factor in success of endodontic treatment. The smear layer contains bacteria, which when left within the canal anatomy may lead to reoccurrence of infection endodontically. When compared to traditional irrigation methods, laser-enhanced irrigation has demonstrated better intracanal smear layer removal.1 As Enterococcus faecalis has been routinely linked to endodontic failures and is a common occupant of the oral cavity, elimination of this bacteria is critical to prevention of reinfection of the canal system. NaOCL as an irrigant has not shown to be effective in elimination of E. faecalis, yet when combined with laser-enhanced irrigation with NaOCL, this bacteria has been eliminated in the canal anatomy.2
Laser-enhanced irrigation

Bulgaria)
Laser energy has been documented to enhance the known effects of NaOCL irrigation through both heating the solution within the canal system and its distant antibacterial effects. But not all laser wavelengths have demonstrated to be equal in effectiveness. The best effects are when NaOCL is combined with an Er:YAG laser as compared to NaOCL alone or when utilized with other type lasers.3 Antibacterial effects were reported to be the best with this combination of irrigant and laser.4 The higher wavelength of the Er:YAG compared to the Nd:YAG or diode was more effective in smear layer removal, hence better at bacterial elimination within the canal system.5 Utilization of a EDTA as an irrigant alternated with NaOCL provides the best debridement of the canal system with enhancement with a Er:YAG laser, as these two solutions have a synergistic effect complementing each other’s effects in the canal anatomy.6
Additionally, the Er:YAG laser (LiteTouch™, distributed in USA by AMD LASERS, Indianapolis, Indiana) creates hydrodynamic pressure following cavitation bubble expansion and collapse when the irrigation solution is activated in the chamber.7-9 Placement of the laser tip does not require entry into the canals to achieve the desired effects, and activation of the irrigation solution in the chamber is sufficient to affect the entire canal system. The LiteTouch™ Er:YAG laser energy is set at a subablative power level that allows its use without structural changes to the hard tissue within the tooth. This eliminates the risks of ledging and perforation of the pulpal floor, allowing safe usage within the tooth.

(Courtesy Prof. Georgi Tomov, Plovdiv, Bulgaria)
When the Er:YAG laser is activated, a heat pulse is generated by the laser radiation delivered via a sapphire tip into an absorbing liquid (irrigant). This results in tensile stress with cavitation being induced in the liquid in front of the sapphire tip at a distance far below the optical penetration depth of the laser radiation. Bubble expansion and collapse cause the surrounding fluid to flow at a speed of up to 12 m/s traveling throughout the canal system. This causes rapid displacement of intracanal fluid via radial and longitudinal pressures sufficient to drive irrigant into the canal anatomy and clean the dentinal tubules significantly. This photomechanical activation of the irrigant includes a temperature rise in the irrigant increasing its effectiveness in debridement of dentinal walls and its tubules and increases the chemical properties of the irrigants.
LiteTouch™-Induced Photo-mechanical Irrigation (LT-IPI ™)
Endodontic treatment is initiated with access to the pulp chamber, which may be performed by traditional methods using burs or by ablation of the enamel and dentin with the LiteTouch™ Er:YAG laser. As the laser is ineffective in removal of ceramics and metals, such as those used in fixed prosthetics and also amalgam, carbides and diamonds are needed create access through these materials. Once dentin has been reached, the laser may be utilized to unroof the pulp chamber (hard tissue mode). An additional benefit of the Er:YAG laser to access the pulp chamber is that it provides decontamination and removal of bacterial debris and pulpal tissue to yield a cleaner chamber aiding its identification of the canal orifices (soft tissue mode).
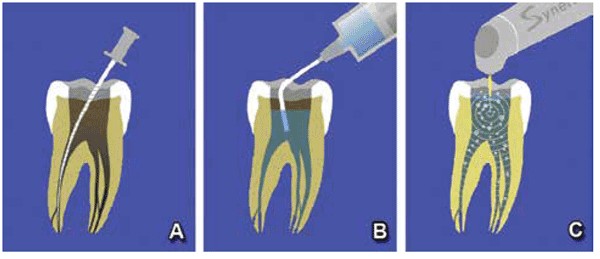
Once the canal orifices are identified, hand files are utilized to establish a glide path to the apical working length in each canal. Canals are then enlarged to the desired ISO canal size with either hand or rotary files (Figure 5A). Laser-assisted canal irrigation requires canal preparation to an apical preparation ISO 25/30 at a minimum. A canal taper of 0.04 or 0.06 for the final instrumentation is recommended. Sodium hypochlorite (NaOCL) is utilized within the chamber and canals during instrumentation both as a pulpal tissue dissolvent and to lubricate the files within the canal, decreasing the potential of file separation that can occur when instrumenting a dry canal (Figure 5B).

Photo-activation of the irrigant within the canal system is performed using the Er:YAG laser with a 0.4/17 or 0.6/17mm tip that assists in removal of the debris created by the files. Between each rotary file, the chamber is filled with NaOCL, and the tip of the laser is placed into the chamber and the solution activated with the laser at 40mJ at 10Hz with an average power of only 0.5W for 20 seconds (Figure 5C). The chamber is suctioned, and fresh NaOCL is placed into the tooth, and the next file is used for instrumentation. It is unnecessary to place the laser’s tip into the canals themselves, as activation of the solution within the chamber transmits down the irrigant into the canals to the apical aspect of the roots. Laser activation may also be performed with 17% EDTA solution alternated with NaOCL. The benefit of EDTA solution is its chelation effect opening canal anatomy so that the next round of NaOCL can reach more pulpal tissue not accessible to the files in fins, as well as accessory and lateral canals. Following final instrumentation of the canals with rotary files, the chamber is filled with NaOCL, and the Er:YAG tip is placed into the chamber again and activated for a minimum of 60 seconds. This allows the photo-activated irrigant to clear debris and remaining pulpal tissue from the complete canal system. The irrigation solution is suctioned from the chamber and fresh irrigant placed and photo-activation repeated until no visible debris (cloudiness) is noted in the chamber fluid. This indicated that all accessible debris has been removed from the canal system. Any remaining solution is suctioned from the tooth, and the canals are dried with paper points. Obturation is then accomplished using the practitioner’s preferred method and materials allowing obturation of anatomy inaccessible by instrumentation with files (Figures 6-7).
Conclusion
The key to endodontic success is two pronged, cleaning the system and sealing it. Although rotary files have improved the efficiency of instrumentation, they are unable to reach any more of the anatomy than hand files are able to reach. Cleaning of the canal system is keyed to irrigation of the canal system to improve debris removal in anatomy that the files are unable to contact. When anatomy is not fully cleaned, sealer is unable to fill this, leaving bacteria to inhabit those areas that may lead to endo-dontic failure over time. Laser-enhanced activation of endodontic irrigants cleans more anatomy adjacent to the main canals so that a more complete obturation of the canal system can occur. An added benefit is that the laser has an antibacterial effect, killing bacteria within the canal anatomy as well as distant to where the irrigation solution may reach essentially sterilizing the entire tooth to the periodontal ligament.
Stay Relevant With Endodontic Practice US
Join our email list for CE courses and webinars, articles and more..


